Translation in the Pharmaceutical Industry

SUMMARY
Medical translation plays a critical role across clinical research, regulatory submissions, and drug marketing in the pharmaceutical industry. WordPar ensures accuracy through expert linguists, certified processes, and multilingual support for over 60 global languages.
The need for accuracy, responsibility and certification in medical translation is or paramount importance. At WordPar, medical translation is undertaken with a scientific approach, right from selection of medical translators to thorough pre- and post-translation checks and reviews.
Clinical Research Translation
India has come to prominence as the world’s pharmacy. Pharmaceutical companies have a multinational framework. Drugs developed in one country are manufactured in another, thanks to specialization and the international supply chain and cooperation. Trials are conducted in several geographical locations and researched data is collected from all over the world. An estimated 80% of clinical trial participants are non-English speakers. Furthermore, drugs manufactured in India and other English-speaking countries are sold to non-English speaking countries.
The fact that stakeholders in the research and supply chain are of different linguistic regions, translation in clinical research and marketing of medicals and pharmaceutical has increased in importance.
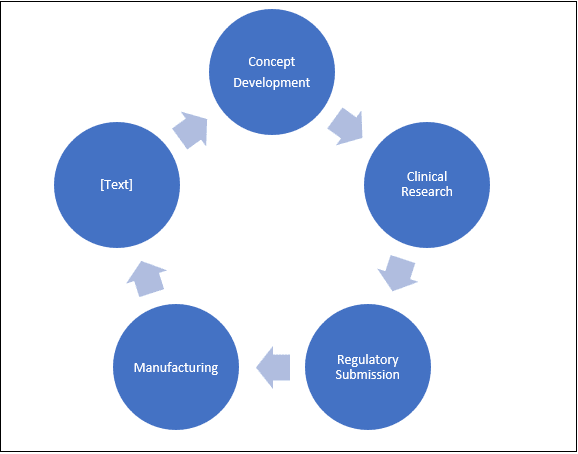
The need for translation arises at various stages of the development cycle of a medicine. Clearly, at the stage of clinical research, the work includes correctly converting data as well as their analysis and reports. The next stage is regulatory submissions, which are required in the respective languages of the countries. Then, at the manufacturing stage, and finally at the marketing and packaging stage, language, communication, and therefore translation plays a vital role.
Approvals are dependent on the authorities of the countries. And countries will require documents submitted in their official languages. Failure to comply with the linguistic requirement can lead to rejection of applications. This has a direct implication of cost escalation, as delay in production and marketing are a loss in revenue. A timely approval helps a company cash in on opportunity and competitiveness that it enjoys at a certain time.
Medical Translation & Localization Services for Drug Companies
All translation in technical subjects requires expertise in the given domain. This principle is of greater relevance in medical translation because it has various implications – medical, regulatory and legal!
Read more about the need for technical expertise in medical translation here.
There are two schools of thought in this regard. One insists that the entire team in the TEP process (translation – editing – proofreading) has medical education, whereas the other proposes that it should be a collaborative process between linguists and medical professionals, and that medical expertise is required at some and not all the stages of translation.
The translation process comprises not only translation, editing and proofreading, but also lexicographical research (generally understood as glossary building) and expert reviews. There are pre- and post-translation stages of the work, in which medical consultants must be involved. The actual translation may be taken up by translators with adequate knowledge of medicine. Translators can be trained on specific terminology and registers and need not be medical professionals themselves.
A linguistic process is replete with domain-specific research, and while it is important to have knowledge of the subject matter, it does not prerequire educational background in the said field.
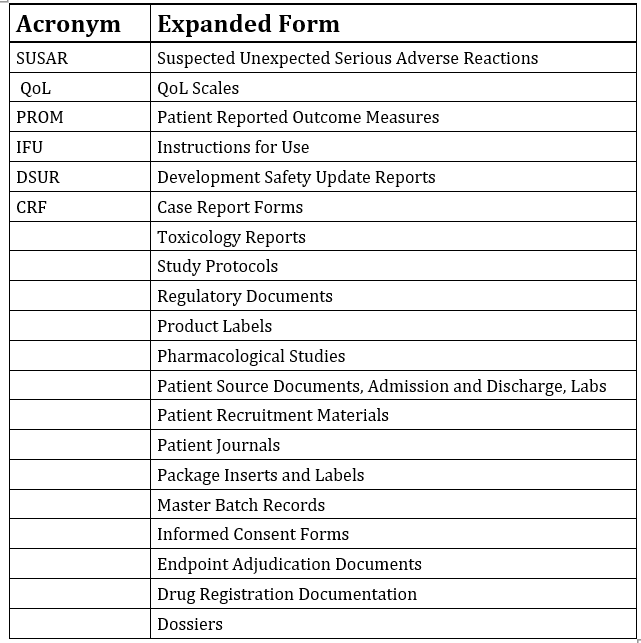
Typical Medical Documentation We Translate for Drug Companies
Below is a list document types that we at WordPar International are in the practice of translating.
We undertake translation of such documents in over sixty languages, not just from and to English, but from and to various languages of the world. These include mainstream languages such as German, French, Spanish, Italian, Portuguese, Dutch, Russian, Hebrew, Mandarin, Japanese, Korean, Arabic, Vietnamese, Thai and Bahasa.
Certified & Notarized Medical Translation
Our team of translators comprise qualified linguists, MD (medical doctors) and nurses. Specialization as a translator in the field of life sciences is a basic minimum qualification in our hiring process. A detailed explanation of our selection process of translators can be found in our blog article here.
Quality controls are stringent, especially in medical translation. Accuracy is assured, and the same is undertaken by our certification of translation and accuracy.
We provide certified translations, which are legally admissible. We also provide notarized and attested copies of the translation as required by the regulations of various countries. This makes WordPar responsible for accuracy and correctness of translation, exonerating the client company of errors.
Backed by authoritative expertise in language, translation, and medical terminology, WordPar’s medical translation team provides complete and well-rounded solutions for the translation needs of the pharmaceutical industry, hospitals, and health care systems.
